|
1/18/2024 0 Comments Nacl ionic or covalent 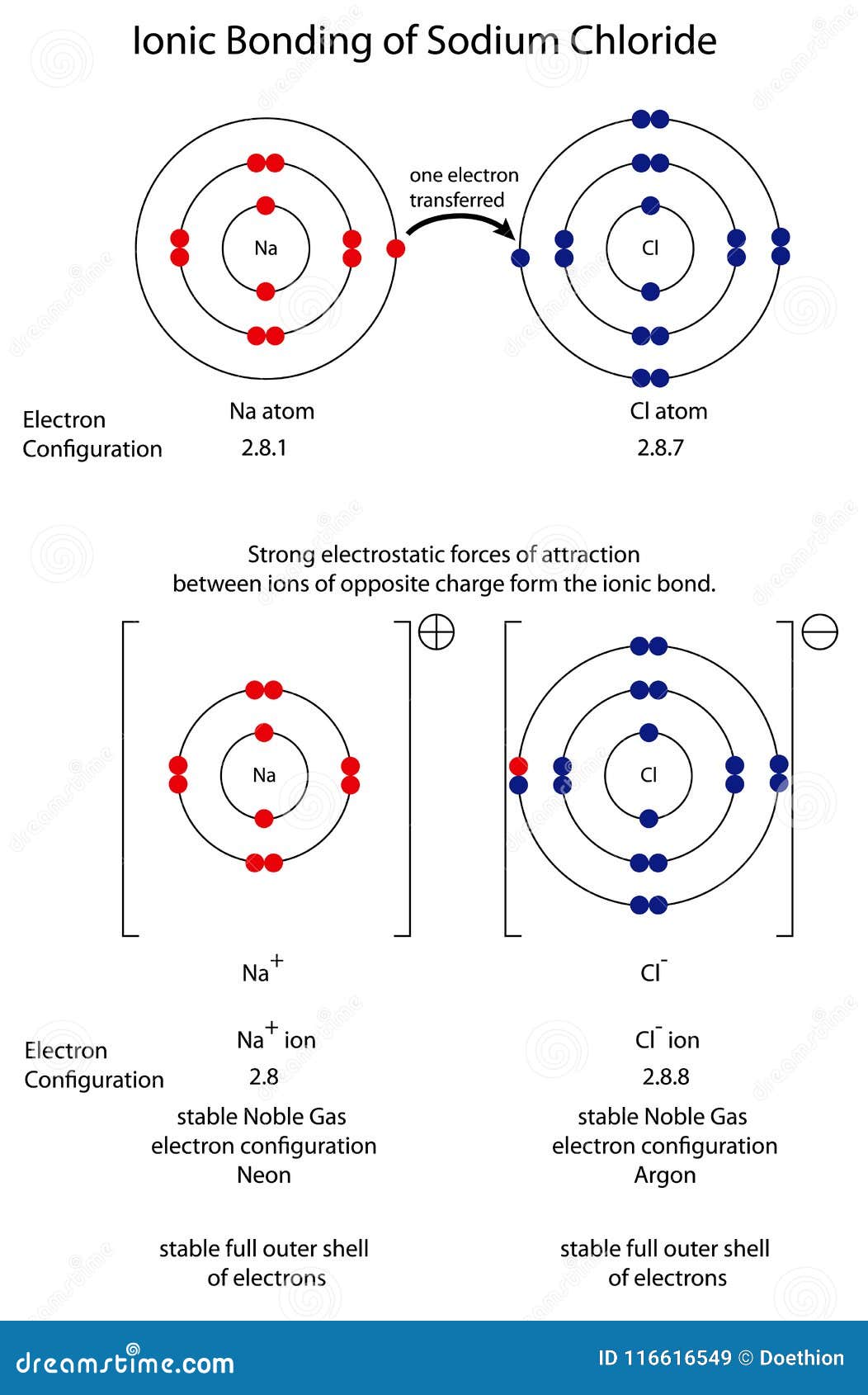
It has only one single valence electron in the 3s subshell.īefore understanding the electron configuration, we will like to introduce the concept of s and p orbitals. The Sodium atom comprises 11 protons as well as 11 electrons. It is important to understand the ionic bond of NaCl. Therefore, with a larger difference in electronegativity, the bond is going to be more Ionic between the two or more atoms. The larger the change of energy the stronger is the bond formation. The overall change of energy plays a vital role in determining the bond. The geometry of the atoms helps in the classification of whether the compound is an ionic or covalent compound. While in the ionic bond the geometry of each atom is determined by the maximum packing rules. The geometry of each atom is determined by the Valence Shell Electron Pair Repulsion Theory (VSEPR Theory) in the Covalent Bond. Such bonds are comparatively stronger than Hydrogen bonds.Īs opposed to covalent bonds where atoms are bonded together by the process of sharing electrons, in Ionic bonds the atoms are bonded by the exchange of two oppositely charged ions. 
In the Ionic bond, the metallic atom loses the electron while the nonmetallic atom gains the electrons. The cations are attracted to the ion pairs on water molecules while at the same time coordinate bonds are also formed.ĭifference between Ionic and Covalent Bond The solubility of the ionic solids in the water will take place depending on the degree of attraction. Several examples have been found where the ionic solids are soluble in water.Īlthough complete solubility doesn’t take place. The bond generally is only based on the electrostatic attraction between the positively charged cations and the negatively charged anions. The main reason being, each of the ions reaches its lowest level of state of energy. Generally, the electron orbitals don’t overlap with each other. Na (+1) and Cl (-1) form an Ionic bond by exchanging electrons. The complete transfer of some electrons in between two atoms or more results in the formation of the Ionic bond.Īn electrostatic force is initiated between the negatively charged anions and the positively charged cations.ĭuring the process of electron transfer, this electrostatic attraction taking place between the two oppositely charged atoms concludes the formation of the Ionic bond. These anions and cations attract each other and form the Ionic bond between the atoms. The transfer of electrons between the atoms results in the formation of positive ions which are known as the cations and also results in the formation of negative ions which are known as the anions. The Ionic bonds are formed between a metal and a nonmetal.Īn Ionic Bond takes place in between two or more atoms, where there is the transfer of one or more than one electron across the atoms. This leads to the formation of Ionic bonds. The ions formed exert electrostatic force upon each other. Therefore, Sodium forms Na (+1) charge, Chlorine on the other hand form Cl (-1). While on the other hand Chlorine, being a halogen forms a monovalent ion by accepting an electron. Sodium being an alkali metal forms a monovalent ion by donating its one valence electron and forms a (+1) charge. So, is Sodium Chloride (NaCl) is ionic or covalent? Sodium Chloride is an Ionic compound, it is formed by the transfer of electrons among its atoms that results in the formation of ions. The crystalline white solid acts as a flame retardant. Sodium Chloride comprises Sodium (+1) which acts as the counterion. There is an abundance of the mineral Sodium Chloride on Earth and is an essential requirement for both plants as well as animals. 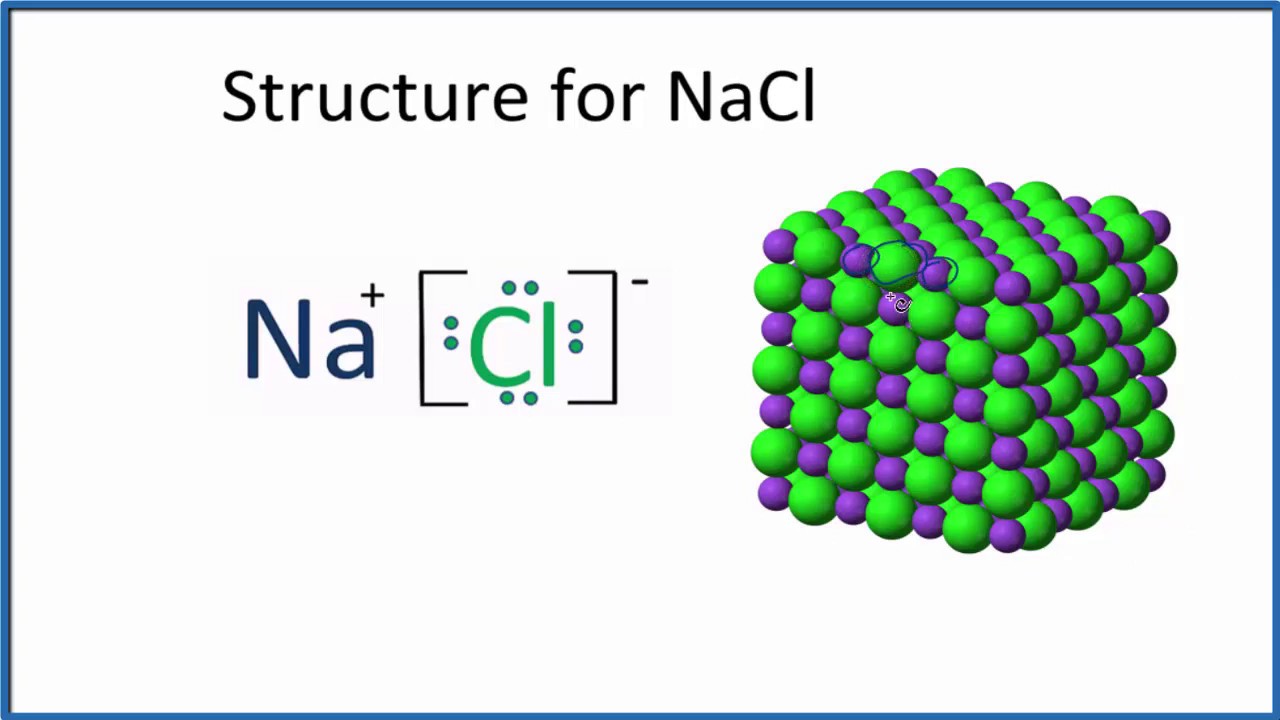
It is also beneficial for the conduction of nerves, contraction of muscles as well as maintaining the proper renal function. As a result of which, in times of need and emergencies the Sodium can be replaced to maintain the intracellular osmolarity. 
Sodium Chloride (NaCl) commonly termed as common salt, is a metal halide that is composed of sodium and chloride.īoth sodium and chloride possess the capability of being replaced. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
